Fumaric acid is a dicarboxylic acid. It has special chemical properties like its molecular structure, acidity, stability, solubility, and reactivity. Scientists know its molecular formula is C4H4O4. This compound looks like a colorless or white powder. It does not have a smell. It tastes fruity and acidic. Manufacturers make about 230 thousand tonnes every year. Most of it is used for food, drinks, and industry. Its physical traits and chemical actions help companies pick it for safe and useful jobs.
Key Takeaways
- Fumaric acid has a simple structure. Its formula is C4H4O4. It is a stable dicarboxylic acid. People use it in many industries.
- It is a weak acid with a pKa of 3.03. This helps lower pH in foods and drinks. It makes their flavor better. It also helps keep them fresh.
- Fumaric acid does not dissolve well in water. It dissolves better in alcohol. This makes it useful in foods that need acidity.
- It has high thermal stability. It can handle high heat. This makes it good for baking and cooking. It does not break down easily.
- Fumaric acid has antimicrobial properties. It fights bacteria in food. This helps keep food safe and fresh.
Chemical properties of fumaric acid
Structure and formula
Fumaric acid has a simple structure. Its chemical formula is C4H4O4. The molecule has four carbon atoms, four hydrogen atoms, and four oxygen atoms. The IUPAC name is (E)-Butenedioic acid. Fumaric acid is the trans-isomer of butenedioic acid. It has two carboxylic acid groups on a four-carbon chain. These groups are on opposite sides of a double bond. This gives fumaric acid a trans shape. This shape makes it different from maleic acid, which is a cis-isomer.
Fumaric acid has a double bond between its carboxylic groups. This bond helps make the molecule stable. It also affects how the molecule reacts.
The general formula for dicarboxylic acids is HO2C−R−CO2H. The R part changes for each acid. Fumaric acid fits this formula because of its special structure. It looks like a white powder with crystals. Its density is about 1.64 g/cm³. The melting point is high, around 287°C. Fumaric acid breaks down before it boils.
| Property | Description |
|---|---|
| Chemical Formula | C4H4O4 |
| Molecular Weight | 116.07 g/mol |
| Appearance | White crystalline powder |
| Density | Approx. 1.64 g/cm³ |
| Boiling Point | Decomposes before boiling |
| Melting Point | 287°C |
| Solubility | Slightly soluble in water; soluble in alcohol |
| Reactivity | Stable under normal conditions; avoid strong oxidizing agents |
| Chemical Stability | Stable in recommended storage conditions |
| Storage Temperature | Store in a cool, dry place away from direct sunlight |
| Vapor Pressure | Negligible |
Acidity and pKa
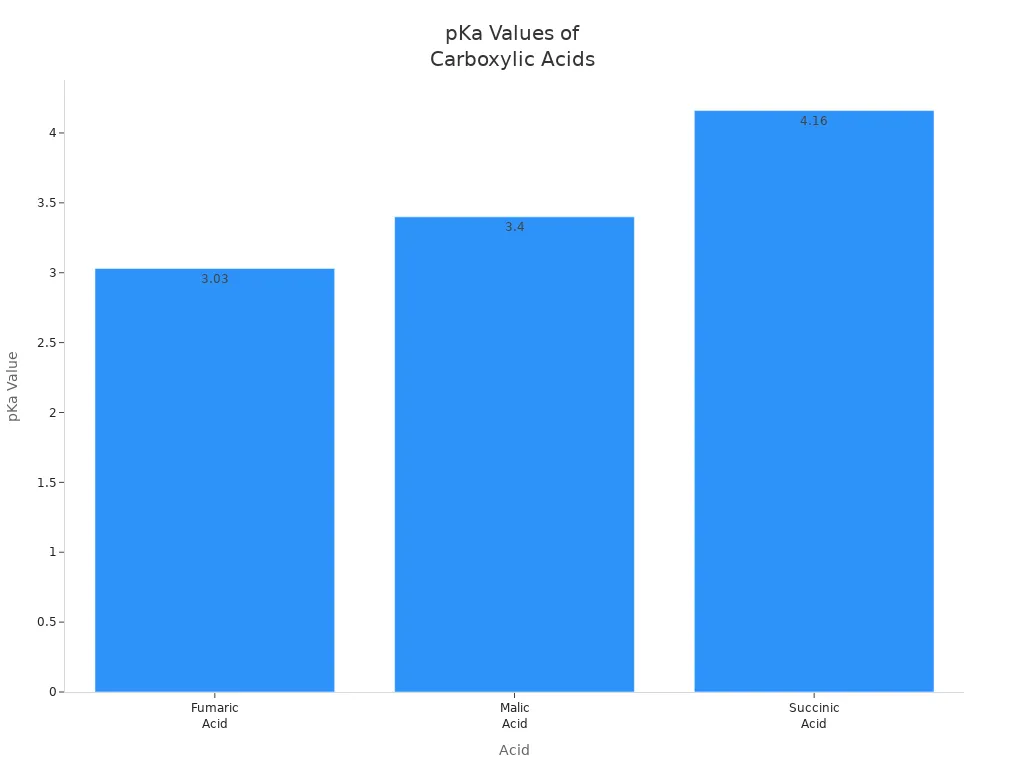
Fumaric acid is a weak acid. It can make solutions more acidic. Its pKa value is 3.03. This means fumaric acid is stronger than succinic acid, which has a pKa of 4.16. It is weaker than malic acid, which has a pKa of 3.40. Fumaric acid helps lower pH in foods and drinks.
| Acid | pKa Value | Acidity Comparison |
|---|---|---|
| Fumaric Acid | 3.03 | More acidic than succinic acid (4.16) but less than malic acid (3.40) |
| Malic Acid | 3.40 | Less acidic than fumaric acid (3.03) |
| Succinic Acid | 4.16 | Least acidic among the three acids |
Fumaric acid can make musts and wines more acidic with small amounts. It does not dissolve well in water. Still, it lowers pH easily. How the acid acts in solutions depends on its hydrophobic nature. This makes it less able to mix with polar solvents.
Solubility
Fumaric acid does not dissolve much in water. At room temperature, only 0.63 grams can dissolve in 100 milliliters of water. If the water is hot, more fumaric acid will dissolve. At 100°C, about 9.8 grams can dissolve in 100 milliliters of water. Fumaric acid mixes better with alcohol than with water. Its hydrophobic nature is why it does not mix well with water.
- Fumaric acid does not dissolve well in water at room temperature.
- It dissolves more as the temperature goes up.
- Fumaric acid mixes better with alcohol and some other solvents.
| Medium | Solubility (g/L) | Comparison to Other Acids |
|---|---|---|
| Water | 5.3 ± 0.2 | ~150 to 230 times lower |
| Musts | 4.7 ± 0.1 | ~150 to 230 times lower |
How well fumaric acid dissolves also changes with pH. At pH 3.5, the uptake rate is highest. At lower pH, more undissociated acid is present.
| pH Level | Specific Fumaric Acid Uptake Rate | Concentration of Undissociated Acid |
|---|---|---|
| 4.0 | Low | Low |
| 3.5 | Maximum | High |
| 3.0 | Increased | Very High |
Fumaric acid’s low solubility does not stop it from being useful. Its strong acidifying power means small amounts can change the pH in foods and drinks.
Stability of fumaric acid
Thermal stability
Fumaric acid can handle high heat very well. This makes it useful in food and factories. Its melting point is between 287°C and 300°C. It does not boil when heated. Instead, it breaks down above 230°C. Most other acids break down at lower heat. Fumaric acid can stay strong at higher temperatures. This means it works well in baking and cooking.
- Melting point is between 287°C and 300°C
- It starts to turn into gas above 200°C
- It breaks down instead of boiling
- It stays stable at room temperature
The structure of fumaric acid helps it fight heat. Its molecules have strong hydrogen bonds. These bonds hold the molecule together when it gets hot. Scientists learned fumaric acid loses its crystal shape only at very high heat. When heated, it changes at certain points. There is a heat peak at 505 K. There is also a shift at 437 K. These changes show when it starts to lose mass and turns into an amorphous form at 513 K.
Chemical stability
Fumaric acid is also very stable with chemicals. It does not break down easily in light or air. Many acids lose power or change color in sunlight. Fumaric acid keeps its properties. This makes it good for products that need to last a long time. Fumaric acid does not react with most chemicals in normal storage. It stays strong in dry and cool places. It does not react with most packaging.
Fumaric acid can handle heat and light better than many other acids.
Fumaric acid’s stability helps it keep its acid power in foods and drinks. It also helps stop spoilage and keeps products safe for longer.
Reactivity and derivatives
Fumaric acid production
Factories make fumaric acid in different ways. One way uses chemical synthesis with maleic anhydride. This method gives a high amount, up to 112% w/w. Another way uses fermentation with glucose as the starting material. Fermentation gives about 85% w/w yield. Glucose is cheaper than maleic anhydride. This makes fermentation better for making lots of fumaric acid. Scientists improved fermentation by using Rhizopus oryzae. They also use metabolic engineering in Escherichia coli. These changes help get more fumaric acid and less waste.
Fumaric acid production is becoming greener. Companies use renewable resources and farm leftovers. This helps lower harm to the environment. Submerged fermentation systems also help fix CO2. This makes the process more eco-friendly.
| Production Method | Environmental Impact |
|---|---|
| Traditional Petrochemical | Causes pollution and global warming |
| Biotechnological (renewable) | Cuts greenhouse gases and fossil fuel use |
Factories need to handle waste from making fumaric acid. Chemical processes make sludge with harmful stuff like naphthoquinone and maleic anhydride. If not cleaned, this waste can hurt soil and water. The process also releases VOCs. These can make air dirty.
Derivative formation
Fumaric acid reacts with other chemicals to make useful derivatives. In labs and factories, it goes through common reactions:
- Bromination happens at the double bond.
- Diesters form when it reacts with alcohols.
- It acts as a dienophile in Diels-Alder reactions.
Fumaric acid makes esters like dimethyl fumarate and diethyl fumarate. These esters are used in medicine and industry. Cross-linked resins from fumaric acid help make unsaturated polyester resin. Salts such as sodium fumarate, calcium fumarate, and potassium fumarate are used to strengthen foods and help medicines.
- Fumaric acid derivatives are used as food acidulants and drink ingredients.
- They work as antibacterial agents in animal feed.
- Polymerization of fumaric acid makes synthetic resins and biodegradable plastics.
- Fumaric acid also helps make l-malic acid and l-aspartic acid. These are important in food and medicine.
Monomethyl fumarate and dimethyl fumarate are used as medicines. Doctors use them to treat psoriasis and multiple sclerosis. The need for fumaric acid derivatives is growing in food, medicine, plastics, and car industries.
Applications and uses
The Acidifying Potential of Fumaric Acid in Various Industrial and Food Applications
Fumaric acid is important in many industries. Food makers use it as a food additive called E297. It gives foods a sour taste and keeps them fresh. Bakers put fumaric acid in dough to control pH and help bread rise. Candy makers use it for a sour flavor that lasts longer. People who make drinks add fumaric acid to improve taste and keep pH steady in drinks and dairy.
“Fumaric acid is the strongest organic food acid used for flavor and pH control. It makes foods taste more sour than citric acid (E330) and malic acid (E296).”
The table below shows how fumaric acid works in different foods:
| Application | Functionality |
|---|---|
| Beverages | Makes drinks taste better and more tart |
| Baked goods | Adds sourness and keeps pH steady |
| Confectioneries | Gives sourness like malic acid |
Fumaric acid can replace tartaric acid. It is found in soft drinks, fruit juices, and sports drinks. It helps make these drinks taste more tart. Factories use fumaric acid to make plastics and other things. It is used in construction, car parts, paints, glues, and sealants.
| Sector | Applications |
|---|---|
| Food and Beverage | – Used as a food additive (E297) for sour taste and keeping food fresh. |
| – Helps bread rise by controlling pH. | |
| – Makes candy taste sour for a long time. | |
| – Improves taste in drinks and keeps pH steady in dairy. | |
| Industrial | – Main part in making plastics with fiberglass. |
| – Used in building materials, car parts, paints, coatings, glues, and sealants. |
- The fumaric acid market may reach US$961.4 million by 2027.
- The industrial part made up about 21% of the market in 2021 and will grow.
- The medicine industry helps the market grow because more drugs and food additives are needed.
Antimicrobial Activity and Its Role in Food Preservation
Fumaric acid helps keep food safe from germs. Scientists know fumaric acid kills bacteria well. Food makers use it to fight germs in apple cider and lettuce. When fumaric acid gets inside a germ cell, it splits into protons and anions. If the pH outside the cell is lower than inside, acid anions build up and slow down germs.
- Fumaric acid splits into protons and anions inside cells at neutral pH.
- Acid anions build up when outside pH is lower, stopping germs from growing.
- Fumaric acid comes off surfaces when wet, lowers pH, and lets anions go into cells.
Studies show fumaric acid works against E. coli, Listeria monocytogenes, and Salmonella. Researchers found that using fumaric acid with ultraviolet-A light kills germs in apple juice.
| Study Title | Pathogens Tested | Findings |
|---|---|---|
| Investigation into the antimicrobial activity of fumarate against Listeria monocytogenes and its mode of action under acidic conditions | E. coli, L. monocytogenes, Salmonella | Fumaric acid worked well against these germs. |
| Inactivating foodborne pathogens in apple juice by combined treatment with fumaric acid and ultraviolet-A light | E. coli O157:H7, S. Typhimurium, L. monocytogenes | Fumaric acid and UV-A together killed germs in apple juice. |
Food safety groups in China, Taiwan, and Australia/New Zealand have strict rules for using fumaric acid in foods. These rules help keep people safe and make sure foods are good quality.
Fumaric acid has special chemical traits that make it useful. It stays strong and does not break down easily. It does not soak up much water. Its strong acidity helps food taste better. It also helps food last longer and stay safe. The table below shows how these traits help in real life:
| Chemical Property | Practical Use |
|---|---|
| Exceptional Stability | Handles baking and heat without breaking down |
| Acidulant & pH Control | Makes food taste good and stops it from spoiling |
| Low Hygroscopicity | Keeps dry foods fresh for a longer time |
Knowing these traits helps people use fumaric acid safely. It is helpful in food, factories, and science.
FAQ
What is the chemical formula of fumaric acid?
The chemical formula for fumaric acid is C₄H₄O₄.
This means each molecule has four carbon, four hydrogen, and four oxygen atoms.
Is fumaric acid safe to eat?
Fumaric acid is safe to eat if you use the right amount. Food safety groups like the FDA and EFSA checked if it is safe.
- It is put in baked goods, drinks, and candy.
- You must follow the rules when using it.
How does fumaric acid help preserve food?
Fumaric acid makes foods more acidic by lowering the pH. This stops bacteria and mold from growing.
Scientists add fumaric acid to apple juice and lettuce to help them stay fresh longer.
Can fumaric acid dissolve in water?
Fumaric acid does not mix well with cold water.
| Temperature | Solubility (g/100 mL) |
|---|---|
| Room Temp | 0.63 |
| 100°C | 9.8 |




